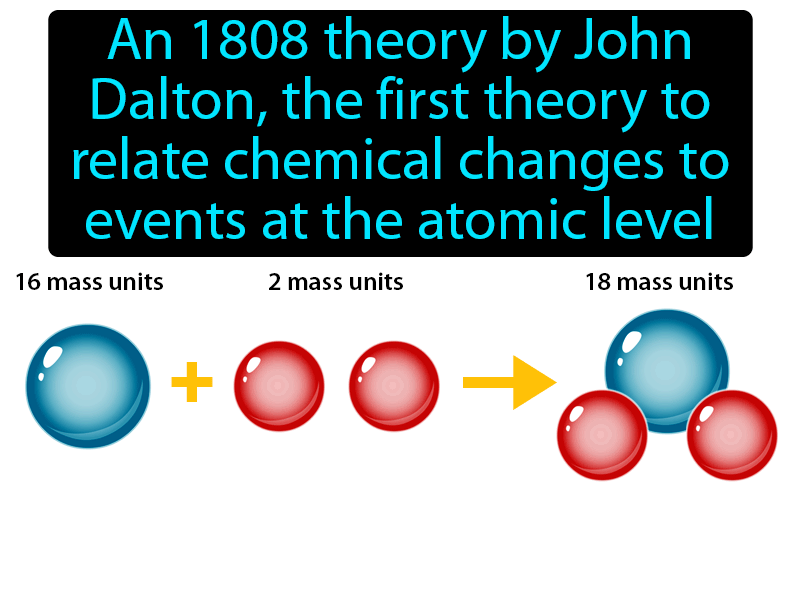
The law of conservation of mass states that the total mass present before a chemical reaction is the same as the total mass present after the chemical reaction. Scientists did not account for the gases that play a critical role in this reaction. More modern atomic theories have made some modifications, for example to include the existence of atomic isotopes, that is, atoms with different numbers of neutrons, but the same number of protons and electrons, and the conversion of energy into matter and vice versa, but Dalton’s core ideas remain valid.\) shows that the burning of word does follow the law of conservation of mass.

Among these rules was the observation that the total matter present in a system does not change during a chemical reaction, although a reaction might lead to a change from a solid to a gas or vice versa. Dalton’s atomic theory stated that different elements were made of different atoms, but did not explain what made the atoms different. Rather surprisingly, Dalton never really explained what led him to propose his atomic theory, although he certainly used it to explain existing rules about how different elements combine. In 1805 Dalton published his atomic theory to explain the observed law of multiple, or definite, proportions, which stated briefly is “when elements combine, they do so in the ratio of small whole numbers”, we will return to this idea later on, in much greater detail. According to modern theory, matter is made of tiny particles called atoms, which are in turn made up of subatomic particles. The first scientific theory of atomic structure was proposed by John Dalton (1766–1844), a self-taught Quaker living in Manchester, England. Atomic theory is a scientific description of the nature of atoms and matter that combines elements of physics, chemistry, and mathematics. That said, it was not until Albert Einstein (1874-1955) proposed his general theory of relativity in 1915 that there was a coherent, mechanistic explanation for gravitational forces. It requires a capacity for deep, original, and complex thought.

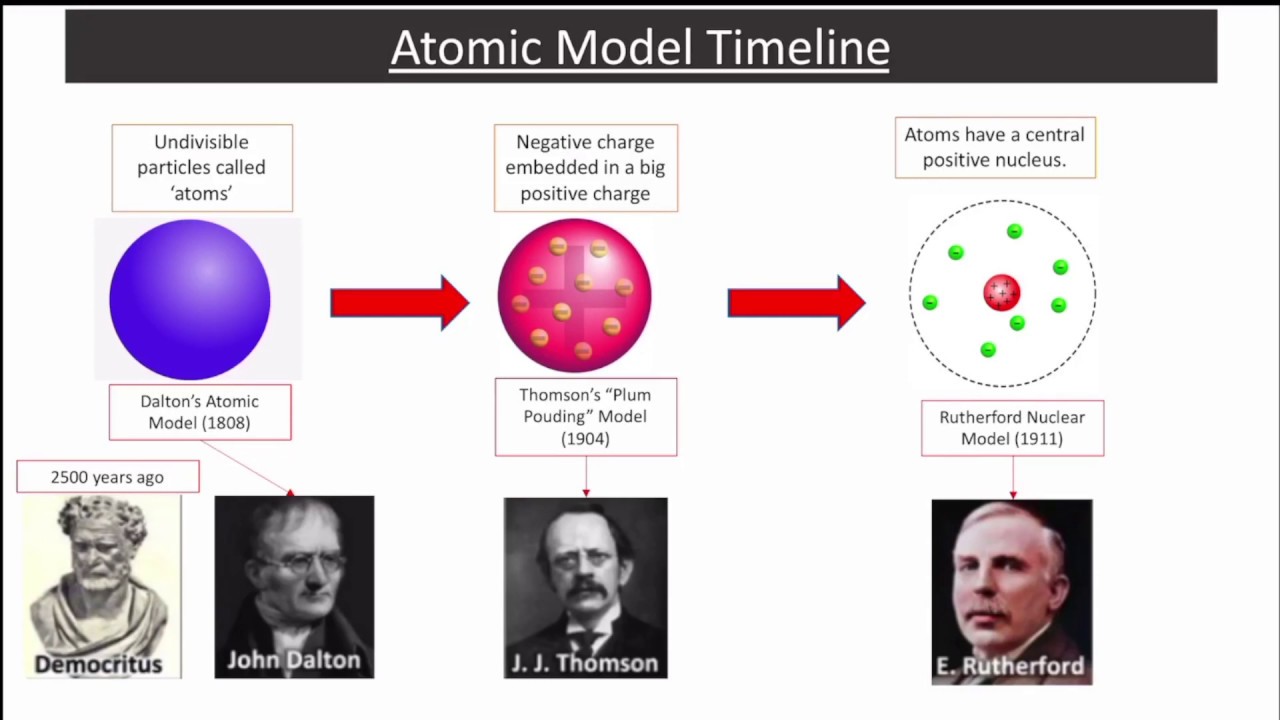
However, the discovery of the existence of subatomic particles (such as electrons, protons and neutrons) disproved this postulate. The point is that Newton was able to make sense of the data, something that is in no way trivial. This theory did not define subatomic particles: According to Dalton’s atomic theory, it stated that atoms were indivisible. This seems like a pretty weird and rather over-blown speculation how does this “action at a distance” between two objects work? Yet, followed scientifically, it appeared to be very powerful and remarkably accurate. \) where \(r\) is the distance between two objects.
